News
The Adrienne Roeder lab at the Weill Institute for Cell and Molecular Biology is contributing insights from plant biology to an interdisciplinary project to integrate green energy into architectural design. Roeder, associate professor in the Weill Institute and School of Integrative Plant Science, Plant Biology Section, in the College of Agriculture and Life Sciences, is collaborating with Jenny Sabin, professor of architecture and design tech in the College of Architecture, Art and Planning, and Itai Cohen, professor of physics in the College of Arts and Sciences, to endow buildings with two traits from the plant kingdom: beauty and diurnal heliotropism, the ability of certain species to turn buds, leaves and other structures to follow the sun from dawn to dusk. Led by Sabin, the project...
Congratulatons to Megan Keller of Doerr Lab for having her story shortlisted in The Scientist’s inaugural science writing publication “What’s Your...
Peptidoglycan (PG) is the main component of the bacterial cell wall; it maintains cell shape while protecting the cell from internal osmotic pressure and external environmental challenges. PG synthesis is essential for bacterial growth and survival, and a series of PG modifications are required to allow expansion of the sacculus. Endopeptidases (EPs), for example, cleave the crosslinks between adjacent PG strands to allow the incorporation of newly synthesized PG. EPs are collectively essential for bacterial growth and must likely be carefully regulated to prevent sacculus degradation and cell death. However, EP regulation mechanisms are poorly understood. Here, we used TnSeq to uncover novel EP regulators in Vibrio cholerae. This screen revealed that the carboxypeptidase DacA1 (PBP5)...
Lilijana Oliver (Roeder Lab), has been awarded a Graduate Research Fellowship from the US National Science Foundation (NSF)! This prestigious fellowship is one of the Foundation’s oldest programs, granting 900 students nationwide fellowships for three-years, endevouring to “ensure the vitality and excellence of the US in science, mathematics and...
Facundo Torres(Doerr Lab), has been awarded a Graduate Research Fellowship from the US National Science Foundation (NSF)! This prestigious fellowship is one of the Foundation’s oldest programs, granting 900 students nationwide fellowships for three-years, endevouring to “ensure the vitality and excellence of the US in science, mathematics and related STEM...
Shuyao Kong (Roeder Lab), is one of only four winners of the 2024 Barbara McClintock award, which is to honor outstanding senior graduate students studying in the Plant Sciences (Plant Biology, Plant Pathology & Plant-Microbe Biology, Plant Breeding, Horticulture or Soil & Crop Sciences) at...
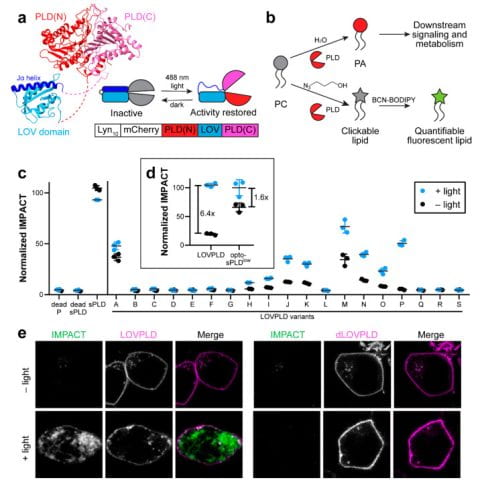
Phosphatidic acid (PA) is a multifunctional lipid with important metabolic and signaling functions, and efforts to dissect its pleiotropy demand strategies for perturbing its levels with spatiotemporal precision. Previous membrane editing approaches for generating local PA pools used light-mediated induced proximity to recruit a PA-synthesizing enzyme, phospholipase D (PLD), from the cytosol to the target organelle membrane. Whereas these optogenetic PLDs exhibited high activity, their residual activity in the dark led to undesired chronic lipid production. Here, we report ultralow background membrane editors for PA wherein light directly controls PLD catalytic activity, as opposed to localization and access to substrates, exploiting a light-oxygen-voltage (LOV) domain-based...
TMEM106B, a gene encoding a lysosome membrane protein, is tightly associated with brain aging, hypomyelinating leukodystrophy, and multiple neurodegenerative diseases, including frontotemporal lobar degeneration with TDP-43 aggregates (FTLD-TDP). Recently, TMEM106B polymorphisms have been associated with tauopathy in chronic traumatic encephalopathy (CTE) and FTLD-TDP patients. However, how TMEM106B influences Tau pathology and its associated neurodegeneration, is unclear. Here we show that loss of TMEM106B enhances the accumulation of pathological Tau, especially in the neuronal soma in the hippocampus, resulting in severe neuronal loss in the PS19 Tau transgenic mice. Moreover, Tmem106b−/− PS19 mice develop significantly increased abnormalities in the neuronal cytoskeleton,...
A cross-college collaboration, led by Paula Cohen and Marcus Smolka, is opening new doors in the study of male infertility by breaking down a key step in sperm formation. Isolating the intricacies of meiotic sex chromosome inactivation, will now enable researchers to identify what happens when that key step...
Endosomes are specialized organelles that function in the secretory and endocytic protein sorting pathways. Endocytosed cell surface receptors and transporters destined for lysosomal degradation are sorted into intraluminal vesicles (ILVs) at endosomes by endosomal sorting complexes required for transport (ESCRT)...
TDP-43 aggregation is a hallmark of neurodegeneration. In this issue, Iguchi et al. (https://doi.org/10.1083/jcb.202302048) report that IκB kinase (IKK), an important mediator of inflammation, phosphorylates cytoplasmic TDP-43 to promote proteasomal degradation, revealing an unexpected link between inflammation and TDP-43...
The location of different actin-based structures is largely regulated by Rho GTPases through specific effectors. We use the apical aspect of epithelial cells as a model system to investigate how RhoA is locally regulated to contribute to two distinct adjacent actin-based...
Crosstalk between auxin and cytokinin contributes to widespread developmental processes, including root and shoot meristem maintenance, phyllotaxy, and vascular patterning. However, our understanding of crosstalk between these hormones is limited primarily to angiosperms. The moss Physcomitrium patens (formerly Physcomitrella patens) is a powerful system for studying plant hormone function. Auxin and cytokinin play similar roles in regulating moss gametophore (shoot) architecture, to those in flowering plant shoots. However, auxin–cytokinin crosstalk is poorly understood in moss. Here we find that the ratio of auxin to cytokinin is an important determinant of development in P. patens, especially during leaf development and branch stem cell initiation. Addition of high levels of...
Postdoctoral researchers Julie Heffler and Avilash Singh Yadav have been awarded the 2023 Sam and Nancy Fleming Research Fellowships by the Weill Institute for Cell and Molecular Biology at Cornell University. These prestigious three-year fellowships support talented young researchers who are doing cutting-edge research in basic biomedical sciences and are planning careers in biological or medical research. “The Fleming Fellowships provide exceptional young scientists the support and freedom to pursue ambitious projects,” said Marcus Smolka, interim director of the Weill Institute and professor of molecular biology and genetics in the College of Agriculture and Life Sciences. “The 2023 recipients are brilliant researchers. Their highly innovative proposals, one on mammalian hearts...
Arf GTPases are central regulators of the Golgi complex, which serves as the nexus of membrane-trafficking pathways in eukaryotic cells. Arf proteins recruit dozens of effectors to modify membranes, sort cargos, and create and tether transport vesicles, and are therefore essential for orchestrating Golgi trafficking. The regulation of Arf activity is controlled by the action of Arf-GEFs which activate via nucleotide exchange, and Arf-GAPs which inactivate via nucleotide hydrolysis. The localization dynamics of Arf GTPases and their Arf-GAPs during Golgi maturation have not been reported. Here we use the budding yeast model to examine the temporal localization of the Golgi Arf-GAPs. We also determine the mechanisms used by the Arf-GAP Age2 to localize to the Golgi. We find that the...
Weill Institute for Cell and Molecular Biology is pleased to announce our 7th Institute Symposium on Cell Signaling and Molecular Dynamics, on October 10th, 2023. We will be joined by 7 distinguished scientists: Tania Baker, Massachusetts Institute of Technology Thomas Bernhardt, Harvard University James Chen, University of Texas Southwestern Chuan He, The University of Chicago Titia de Lange, The Rockefeller University Seung Yon (Sue) Rhee, Carnegie Institute for Science Sara Wickstrom, Max Planck Institute for Molecular Biomedicine This year, we are also thrilled to present the inaugural Emerging Scholars Symposium on October 9th, 2023. The Weill Institute has selected 8 exceptional senior Ph.D. students in the life sciences, from institutions...
Eight graduate students from across the U.S. to attend inaugural Weill Institute Emerging Scholars Symposium in Oct....
Dr. Adrienne Roeder’s study of polyploid cells in the mustard-like plant Arabidopsis thaliana was featured in the August 2023 issue of Science News. By mutating genes that control polyploidy, she and her team determined that polyploid cells are the key to proper flowering. If the plant sepal had too few polyploid cells, it was stiff and upright and blooming was impeded. Too many such cells caused the sepals to splay and expose the petals prematurely. Roeder and her postdoc Batthula Vijaya Lakshmi Vadde discovered that normal, diploid cells turn into polyploid versions when a protein, ATML1, kickstarts the process, in part by stimulating the production of certain fatty acids. Roeder’s graduate student Frances Clark has found that polyploidy, spurred by the same molecular signals,...
Brittany White-Mathieu, an alumna of the Baskin Lab, was the recipient of a Blavatnik Regional Award for Young Scientists for advancing the field of chemical imaging to further our understanding of lipids, and their role in cellular function and disease, including cancer. She has created a revolutionary method, Lipid Expansion Microscopy, that enables super-resolution imaging of lipids within cells using widely available instrumentation. White-Mathieu’s work enables direct study of these compounds in cellular membranes where critical cell signaling events and nutrient exchange occur. White-Mathieu will begin a new faculty position at the University of New Hampshire in late August...
When a cell divides, faithfully replicating its DNA is essential – the life of the cell is on the line. This year’s recipient of the 2023 Harry and Samuel Mann Outstanding Graduate Student Award, Jumana Badar, is carrying out research to determine how cells ensure DNA is replicated correctly, a question that is central to how healthy cells reproduce and how drugs that make this process more difficult for the cell could be optimized to target cancer cells. , Click to open gallery view Credit: “I am honored and humbled to be chosen as this year’s recipient,” she said. “This award will enable me to seek out opportunities to gain new skills and exercise the ones I already have.” When cells replicate, the DNA strands must separate from one another to form what is called a...